Deterministic and probabilistic human health risk assessment for fluorides in drinking groundwater from Lukavac, Bosnia and Herzegovina
Article information
Abstract
Although fluorides are essential for dental health, there are growing health concerns regarding the risk-benefit ratio of fluoride exposure. The objectives of the study were to obtain data on the amount and human health risks of fluoride in drinking groundwater, as well as to compare and evaluate the differences in the outputs obtained by two different approaches in health risk assessment (deterministic vs probabilistic). From a densely populated industrial area of northeastern Bosnia and Herzegovina, a total of 54 groundwater samples were collected. Fluoride concentrations varied from 1.69 to 3.52 mg/L. The WHO’s threshold value for fluoride in drinking water was exceeded in all the samples analysed, indicating an increased daily intake of fluoride from groundwater. Deterministic and semi-probabilistic techniques were used for exposure assessment and health risk quantification. Generally, the deterministic approach resulted in acceptable health risks in most adult exposure scenarios. However, the Monte Carlo simulation revealed that 20.6, 20.8, and 99.8% of adult males, females, and children, respectively, were above the tolerable upper intake level, indicating that both adults and children face a significant health risk. Nevertheless, small children are more vulnerable to environmental hazards than youth and adults. Hence, a more in-depth risk-benefit analysis is required to reduce/or optimize fluoride content in drinking water to prevent tooth decay and fluorosis at all ages. Considering that optimal daily intake of fluorides is a crucial factor for preserving human health, decision-makers should take steps to emphasize the importance of continuous monitoring of fluoride concentrations in drinking water.
Introduction
A sufficient amount of clean and uncontaminated drinking water is a fundamental prerequisite for human life on the earth. The need and the consumption of drinking water are constantly increasing, leading to the inability to balance in between them. In addition, the increasing water pollution leads to a significant lack of clean and safe water intended for human use [1].
Both natural and anthropogenic activities contribute to drinking water pollution. Although the water presence of certain chemical elements is vital for human when present at concentrations above the maximum permitted levels, they can adversely affect human health. Environment contaminated with salts referred to as fluorides has been recognized as one of the major problems worldwide due to the particular interest in human health [2]. Fluorides are present in water, air, groceries, soil, dust, rocks, and personal hygiene products such as kinds of toothpaste and mouthwashes [3–10]. Also, their widespread use as insecticides and rodenticides lasts for decades [11–13]. Although fluorides are essential in preventing dental decay [14–17], rising health concerns are accounted for by increased groundwater pollution and the intensive fluoridation of drinking water.
Fluoride is found in groundwater and surface water, mainly due to the stream through the soil and rocks. Industrial plants discharge and inadequate household waste disposal into the environment could cause an additional burden on the groundwater. Fluoride contamination in groundwater has been considered a severe health and environmental problem worldwide [3]. Consequently, many countries in the world have already stopped the process of intensive drinking water fluoridation. Currently, there is no practice of adding fluoride to the tap water in Bosnia and Herzegovina. Furthermore, Arslanagić Muratbegovic et al. [18] defined this location as having less than 0.1 ppm fluoride naturally present in the drinking water.
Bosnia and Herzegovina is a developing Southeast European country where intensive urbanization and industrialization cause negative environmental repercussions and contribute considerably to groundwater pollution. It is common practice for the residents to use groundwater from public water sources for household needs (drinking, preparing food, etc.) without any previous water quality analysis. According to a previously published study, fluorides in groundwater have been reported to range from negligible values to over 25 mg/L [19]. Fluoride at both low and high concentrations can adversely affect human health. The toxicity of high dose of fluoride is mainly related to developmental disorders in teeth and bones known as dental and skeletal fluorosis. Excess fluoride ingestion over a prolonged time may also lead to dysfunctions of many organs including the thyroid gland [20], kidneys [21], brain [22], stomach [23], heart [24], etc. Epidemiologic data suggest that children below the age of 6, as well as children during fetal development, are at higher health risk of experiencing the toxic effects of fluoride poisoning due to elevated exposures from water in fluoride endemic areas [24,25].
Therefore, the study aimed to quantify the fluoride content in drinking water collected from 18 public water sources from Tuzla Canton, Bosnia and Herzegovina. Fluoride content was also assessed in terms of its health effects. Traditional deterministic and modern probabilistic approaches in human health risk assessment were used to estimate potential risks from fluoride ingestion through drinking water for adult males, females, and children separately, as well as to compare the results of two different risk assessment methodologies. The study was designed to provide the first official baseline information on drinking water safety in terms of fluoride content for residents and decision-makers. These findings are intended to be valuable for future planning of the water fluoridation process, as well as in the process of human health protection.
Materials and Methods
Study area
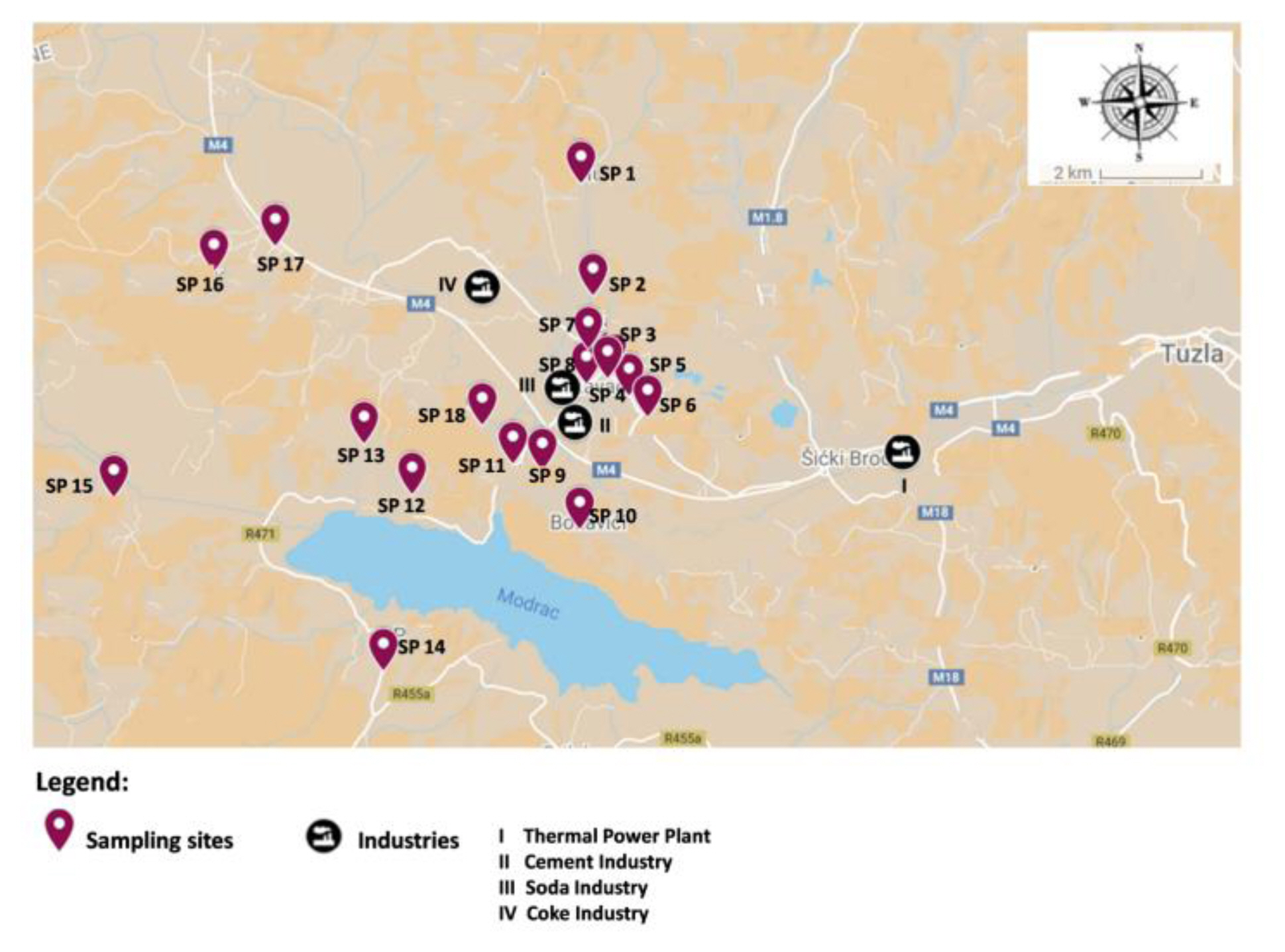
The Lukavac city (44°33′N, 13°31E), which has a population of 44.52 thousand people, is in the Tuzla Canton in Bosnia and Herzegovina’s north-eastern region. Lukavac is a town with an industrial character. The main factories in its vicinity are Soda Lukavac, cement factory, Global Ispat coke industry, Thermal Power Plant (TPP) Tuzla, and several brown coal and lignite mines. The supply of tap water is ensured by direct pumping of water from Modrac Lake. Modrac Lake is an artificial lake that is largely located in the Lukavac, but it partly borders the towns of Tuzla and Zivinice. This area is rich in numerous natural sources of drinking groundwater. Here, bottled spring water and groundwater from public water sources are frequently used for household needs due to concerns about the quality and safety of tap water.
Water sampling
Water samples were collected in triplicates from 18 locations. The methodology of water sampling was carried out according to the standard procedures ISO 5667-3 [26] and 5667-5 [27] in the spring of 2021. Each sample was collected in a volume of 500 mL in pre-cleaned glass sampling bottles resilient against chemicals and analysed the day after. All samples were collected on the same day when there was no rainfall or other inclement weather. The sampling locations are indicated in Figure 1.
Sample analysis
The pH/mV meter MA 5741 (Iskra, Slovenia) with fluoride ion-selective electrode (Boeco, Germany) and magnetic stirrer MMS 3000 (Boeco, Germany) was used for the determination of fluoride in drinking water. All reagents were purchased from commercial sources and were of analytical grade purity. Fluoride stock solution (0.1 mmol/L) was made by dissolving sodium fluoride in deionized water. Before use, the prepared stock solution was diluted to obtain 10 mmol/L working standard solution. Afterward, in order to get a series of measuring standard solutions, the previously prepared working standard solution was diluted with deionized water. A total ionic strength adjustment buffer (TISAB) in a ratio of 1:1 was added to the prepared measuring standard solutions in a polyethylene dish. It was made of sodium chloride (58 g), glacial acetic acid (57 mL), sodium citrate (300 mg), and distilled water up to 500 mL. Sodium hydroxide (5 mol/L) was used for adjustment of pH to the value of 5–5.5. Subsequently, the buffer was diluted with water to the volume of 1L and stored in the polyethylene bottle [10]. A calibration curve was prepared using analytical standards. The analytical method for fluoride determination in water was linear in the range of 25 μmol/L to 400 μmol/L.
The temperature of the water samples was balanced to room temperature before the analysis. A subsample of 10 mL water was mixed with 10 mL of TISAB buffer in a polyethylene beaker. Adding TISAB buffer in the sample prevents the formation of complexes of polyvalent cations with fluorides, thus eliminating the expected interference. The fluoride ion-selective electrode tip was immersed into the prepared solution. The potential difference was measured separately for each sample and its triplicate. The obtained results were expressed in μmol/L and converted to mg/L.
Exposure assessment and risk characterization
In risk assessment, both deterministic and probabilistic approaches were applied. To assess human’s fluoride exposure, the estimated daily intake (EDI) was calculated as follows:
where EDI is the estimated daily intake of fluoride through water ingestion (mg/kg/day), C is the concentration of fluoride in the water (mg/L), and IngR is the ingestion rate of water (L/day).
Risk characterization was quantified under the current environmental circumstances in Lukavac city. The human non-cancer risk was calculated using equation (2):
where, HQ is hazard quotient (unitless), and RfD is the oral reference dose of fluoride (mg/kg/day). If the EDI is less than the RfD, HQ≤1, the risk is considered to be acceptable. If the EDI exceeds the RfD, HQ>1, it is likely that there will occur some adverse effects on human health [28].
Monte Carlo simulations
Probabilistic risk assessment is a computerized mathematical technique based on Monte Carlo simulations. The input variables are not fixed values nor sampled randomly, and the output is presented as a probability distribution. To obtain information about how likely each outcome is @RISK 5.5 software (Palisade Corporation, USA) was used. A randomly selected input variable allows over and over again computing different outputs based on the assigned equation. An optimal number of iterations to be run was set to 10,000. Based on a single simulation performed, mean, maximum, 75th, and 95th percentile values of EDI and HQ were derived.
Exposure scenarios
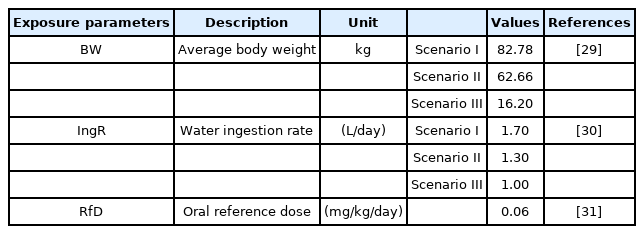
Based on the physiological and behavioural differences, the population was divided into three groups. Scenario I and II included adult males and females, respectively, while Scenario III included child residents of the Lukavac area. All input parameters are presented in Table 1 and Table 2.
Statistical analysis
All experiments were set up in triplicates and the results were expressed as mean values. The data were analysed using a statistical package IBM SPSS Statistics (version 21).
Results and Discussion
Fluoride is released into the air, soil, and water during natural and anthropogenic activities, among which volcanic eruptions, underground coal combustion, mining, and various industrial processes contribute the most. A particular problem for groundwater contamination with fluoride is a discharge of phosphate fertilizer into the environment. Coal-burning is a substantial cause for increment in human exposure to fluoride released as fumes. In areas where indoor coal utilization is frequent, numerous residents show typical signs of fluoride intoxication [32]. Daily combustion of enormous quantities of fossil fuels, in particular, brown coal and lignite by the TPP Tuzla and individual household furnaces as well as industrial emissions, and improper disposal of waste from factories and households release pollutants into the environment of the Tuzla Canton [33–35]. However, drinking water, foodstuffs, and fluoride-containing dental products are currently considered major contributed products to the increased fluoride intake in the human organism. Among the above, most of the scientific evidence confirms the fact that drinking water is the main and major source of human exposure to fluorides [7, 24, 36–40]. The amount of fluoride in groundwater is determined by heterogeneous factors, including the soil through which the water leaches, agricultural production practices, the proximity of factories, the type of water pipes used, the population of the area, and many more. As we previously highlighted, our study area is characterized by numerous polluting industries, including TPP, a coke factory, a cement factory, and several mines [29]. Considering that there is a lot of agricultural land in their vicinity and that people often use fertilizers in agricultural production, which mainly contains fluorine in defined concentrations, fluoride contamination of groundwater could be due to the soil leachate [41].
The current study was carried out to provide basic information on fluoride content in groundwater intended for human use. Although a previously published study from the nearby emphasize that the Tuzla Canton faces low fluoride content in drinking water [42], our findings do not confirm it. According to the analysis of 54 drinking groundwater samples, fluoride concentrations ranged from 1.69 to 3.52 mg/L with a mean concentration of 2.44 mg/L (Table 3). Obtained concentrations were compared to the World Health Organization’s (WHO) quality guideline value for fluoride in drinking water [41]. The results revealed that all analysed samples exceeded the WHO’s 1.5 mg/L recommended threshold. Although our data show higher fluoride levels than suggested, they are still within the allowable concentration range compared to the US Environmental Protection Agency’s (US EPA) rules, which set the maximum fluoride content in drinking water at 4.00 mg/L [43]. Apparently, two eminent institutes appear to have significantly different numbers for the maximum allowed fluoride concentration in the water intended for human use. It can lead to misinterpretation of the results. However, even if the obtained fluoride concentrations are below the US EPA’s standard, there is a reasonable cause for concern, especially given that excessive fluoride exposure in drinking water, alone or in combination with exposure from other sources, can give rise to several adverse effects.
In a large meta-analysis and a systematic review of data in India, it was found that fluoride is present in groundwater in concentrations around 2.37 mg/L with a 95% confidence interval [44]. While Bosnian Herzegovinian groundwater is not classified as water polluted by fluoride, it is well known that India is one of the countries that continuously face the problem of fluoride polluted groundwater. In a 2019 Indian study, Yadav et al. [45] analysed 28 groundwater samples, with about 64% of the samples recording fluoride content above the WHO’s threshold value. The average concentration of 1.88 mg/L was obtained, which contributed that 29% of adults and most infants and children face an increased non-carcinogenic health risk. It is a well-known fact that underdeveloped and developing countries face the greatest challenges of fluoride groundwater pollution. In addition to India, this problem has been recognized in endemic regions in Iran [46], Pakistan [47], Africa [48], Ukraine [49], where fluoride was found in groundwater samples in concentrations of 1.8 mg/L, 2.0 mg/L, 1.95 mg/L, and 2–7 mg/L, respectively.
It is well documented that fluoride present in slightly higher concentrations than recommended can cause various health problems, including endocrine and neurological disorders, renal dysfunction, and health issues with bones and teeth causing skeletal and dental fluorosis [26, 40–46]. Recent literature reports that fluorosis is a global problem affecting more than 70 million people in 25 countries. It is estimated that more than 200 million people worldwide rely on drinking water with fluoride concentrations that exceed the WHO’s maximum permitted limit [47]. All of these pointed to the necessity for a more comprehensive health risk assessment of fluoride exposure in Lukavac’s drinking water. The non-cancer health risk was assessed using a deterministic point estimate, as well as a probabilistic technique that took into account variability and uncertainty (Table 4).
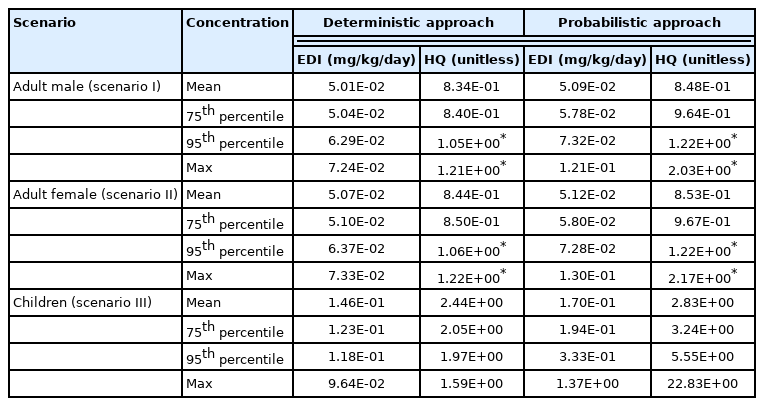
Summary of estimated daily intake of fluoride and non-cancer risk values for two exposure scenarios based on the deterministic and probabilistic approach.
Deterministic risk characterization is calculated based on mean, 75th, 95th, and worst-case (maximum) values for two input variables (BW and fluoride concentration). As shown in Table 4. the deterministic risk assessment indicates that child’s consumers are at considerable non-carcinogenic risk. In our study, the assessed risk was significantly higher in children than in adults. It has been reported that due to low body weight, children are more exposed to fluorosis [47]. Another reason is that fluorides from water can faster and at a greater rate accumulate in metabolically active and vascular bones and teeth of children [50]. The mean estimated daily dose for children was 0.15 mg/kg/day. Our results suggest that it is not safe for children of Lukavac to consume groundwater, considering that the HQ value for children below 6 years of age in all scenarios was greater than 1. These results are similar to the results reported in studies conducted in India [44,45]. Yousefi et al. [38] performed the deterministic health risk assessment of fluoride through ingestion of groundwater for residents of Poldasht city, West Azerbaijan Province. They divided population into four age groups as fallow: <2 years, 2–6 years, 6–16 years, and ≥16 years. Their results indicated that children aged 2–16 years are at a more severe non-carcinogenic health risk than infants and adults. They highlighted children aged 2–6 years as a hyper-sensitive population to fluoride exposure. The findings were similar to the study from Pakistan [51]. They also reported that children were at higher risk of fluorosis as compared to the elderly. Our results are complementary to the existing group of evidence focused on greater sensitivity and vulnerability of the child population compared to the adults [44,45,51]. As shown in Table 4. the results for adult males, based on mean and 75th values, for which intakes of 5.01E-02 mg/kg/day and 5.04E-02 mg/kg/day were calculated, suggested that the health risk of fluoride exposure is acceptable. When the 95th and maximum values were applied, the resulting HQs indicated increased health risk. Similar to the adult male scenario, when the two most adverse exposure scenarios for women were applied, the increased health risk from an intake of 6.37E-02 mg/kg/day and 7.33-E02 mg/kg/day fluoride via drinking water was quantified. The purpose of this worst-case approach in health risk assessment was to obtain a simple answer as to whether the results obtained indicate ‘safe’ or ‘not safe’ use of the analysed water, as well as to ensure that all exposed people are protected under all possible circumstances [35, 52]. A lack of reliable information about which input variables contribute to greater or lesser safety levels is evident in the deterministic approach in health risk assessment. To overcome this problem, a more exhaustive probabilistic technique was applied.
Distribution modelling is carried out with @RISK software allowing us to obtain more detailed and reliable statistics on the percentage of the exposed population at increased health risk. Table 4 shows a summary of the findings. Ingestion of fluoride contaminated water by adult males, adult females, and children resulted in mean risks of 8.48E-01, 8.53E-01, and 2.83E+00, respectively, based on the cumulative probability distribution of values, indicating that approximately 8.48–8.53 adults per thousand people, and 2.83 child residents per hundred children will experience adverse effects from excessive fluoride intake.
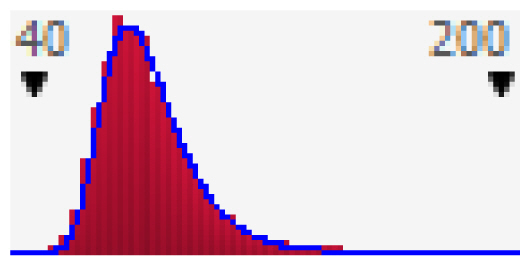
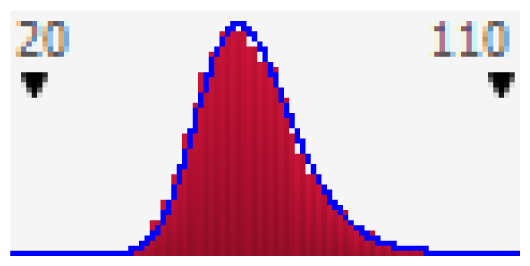
Since the exposed population in this study was divided into three groups, the probability distribution curves of HQs were presented separately (Figure 2). If HQ is higher than 1, a considerable adverse effect could be observed. The results showed that the increased health risk of adverse effects due to fluoride water ingestion is expected in 20.6% of the male, and 20.8% of the female population. According to the results of both approaches in risk assessment, females are slightly more vulnerable to intake of fluoride from water per unit body weight than males due to their physiological and behavioural differences (e.g., body weight, water ingestion rate, etc.). Based on the analysis of published studies, we found that the deterministic method was primarily used to assess the health risk of fluoride exposure. One of the few studies based on Monte Carlo modelling is the Fallahzadeh et al. [39] study conducted in 2018 in Iran. They divided the population into three groups as follows: 3–10 years, 11–20 years, and 21–72 years of age. A probabilistic risk assessment was performed on 1,000 iterations using Crystal Ball software. The obtained results are comparable with the results of our study. In Meybod county, which was the most polluted county in Iran, the estimated risk based on HQ values for the mean values in both teens and adults were less than 1 and for children was higher than 1, indicating a high non-carcinogenic risk for the children age group [39]. In the current study, we found that the mean HQ value in the child population is around 3.3 times higher than in adults. Based on the cumulative probability curve 99.8% of children are exposed to the risk of excessive fluoride intake. To determine the most important variable affecting the increase in health risk in children Fallahzadeh et al. [39] performed an additional sensitivity analysis. According to the results, the drinking water ingestion rate seems to be the most influential variable in the health risk assessment in the child population.
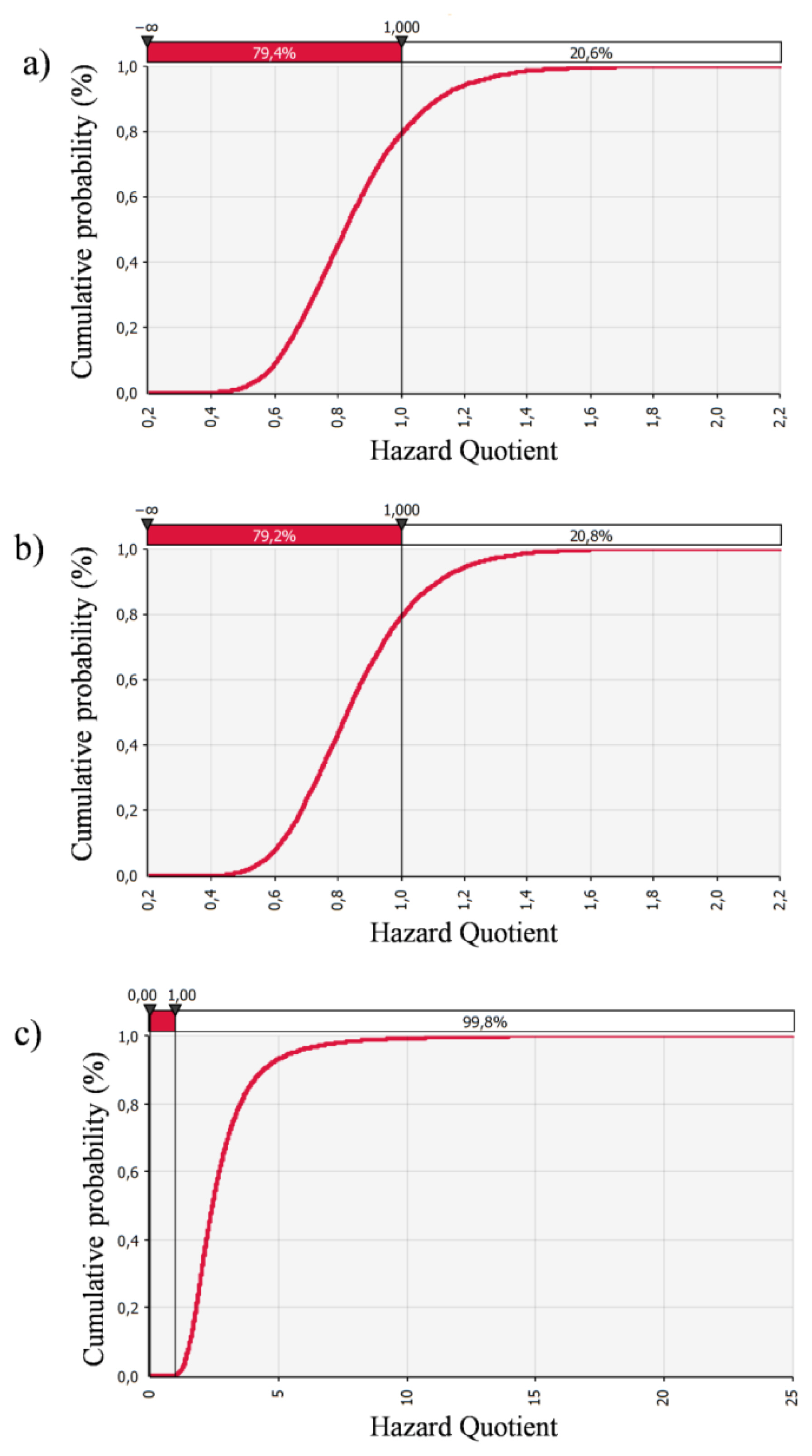
Cumulative probability plot of HQ in the adult male (a); adult female (b); and children (c); groups due to ingestion of water content of fluoride.
At an intake of 0.3 mg/kg/day, the first indications of acute fluoride intoxication appear [39]. In our study, given that the EDI for Scenario III at the 95th percentile was 0.33 mg/kg/day, the signs of acute fluoride intoxication in children could be expected.
Conclusions
In the present study, we evaluated drinking water safety by assessing the non-carcinogenic human health risk using a conservative deterministic and structured probabilistic approaches. Although the deterministic risk assessment procedure relies on different underlying assumptions and concepts compared to the probabilistic method, we found that both techniques can provide similar results. An additional problem could arise in the child population. Children are more vulnerable to all kinds of environmental pollutants than adults. However, conducting deterministic risk evaluation alone may not be sufficient to adequately assess health risks due to the possibility that the health risk in different studies could be either under- or overestimated. Hence, it is required to estimate the risk by using probabilistic simulations. The Monte Carlo simulation can generate enough random values and predict all possible outcomes presenting them as the probability functions.
Both approaches gave similar results, indicating that, in worst-case scenarios, human health could be jeopardized by fluoride exposure through groundwater. Furthermore, probabilistic risk analysis revealed that more than 20% of the observed adult population and almost 100% of the child population are at increased health risk from excessive fluoride intake. However, the estimated health risk is not an ultimate measure of overall adult health risk. One of the main limitations of the current study was that commercially bottled water, which is also frequently used for drinking, was not considered or analysed. In addition, toothpaste and mouthwashes, food, air, etc. contribute to overall fluoride exposure as well. Therefore, it’s reasonable to predict that using an aggregative approach to risk assessment would result in a significantly increased health risk. This preliminary study highlighted the need of conducting a more comprehensive study to evaluate the risk more accurately to human health in the study area. Concurrently, continual decision-making activities, including public informing, are needed to optimize the fluoride exposure of inhabitants.
Notes
The authors have no conflict of interest to declare.
CRediT author statement
MŠH: Conceptualization, Methodology, Software, Formal analysis, Resources, Data curation, Writing-Review & Editing, Supervision; NH: Validation, Investigation, Resources; HM: Data Curation, Writing-Original draft Preparation, Data curation, Visualisation; AT: Formal analysis, Investigation, Writing-Original draft Preparation, Visualisation; AH: Resources, Data Curation, Writing-Original draft Preparation, Funding acquisition.