AbstractTitanium dioxide (TiO2) was used in various applications in a wide range of products including food, cosmetics and photocatalyst. General toxicity studies of titanium dioxide, GST (Green Sludge Titanium) have been investigated in several reports, whereas studies concerning mutagenicity and genotoxicity have not been elucidated. Herein, we investigated the potential mutagenicity and genotoxicity of GST by genetic toxicology testing. The bacterial reverse mutation test was conducted by the pre-incubation method in the presence and absence of metabolic activation system (S9 mixture). The chromosome aberration test was performed using cultured Chinese hamster lung cell line in the absence and presence of S9 mixture. The micronucleus test was performed by using specific pathogen-free male ICR mice. Genotoxicity tests were conducted following the test guidelines of the Organisation for Economic Cooperation and Development with application of Good Laboratory Practice. No statistically significant increases were found in the bacterial reverse mutation test, in vitro chromosome aberration test, and in vivo micronucleus test when tested for induction of genotoxicity in GST. These results suggest that GST did not induce mutagenicity and genotoxicity in both in vitro and in vivo system.
IntroductionTitanium dioxide (TiO2) are among the most widely produced and used photocatalyst in the world [1]. Titanium dioxide (TiO2) is an oxide of titanium that exists in nature in different crystalline forms; the most common form is rutile, whereas anatase and brookite are comparatively rare forms. These materials possess some unique properties for which they are used in many products; however, these same properties could also be the source of potential harm to biological systems, causing adverse effects [2]. In places like laboratories or industries where these TiO2 are produced, handled or processed, the workplace exposure can occur. Workers involved in TiO2 production can be exposed to TiO2 dust, the human body is exposed to TiO2 through four possible routes: inhalation, ingestion, skin penetration, and injection of nanomaterials as in nanomedicine [3]. A review of TiO2 genotoxicity data summarizing both in vivo and in vitro results was last published in 2014 [4]. In the present article, we have gathered and discussed the recent genotoxicity data of TiO2 and in vitro and in vivo genotoxicity studies are summarized.
The aim of this study was to determine the potential mutagenicity and genotoxicity of titanium dioxide, GST (Green Sludge Titanium). For accurate evaluation of genotoxicity, three types of in vitro and in vivo genotoxicity tests, i.e., the bacterial reverse mutation test, in vitro chromosomal aberration test, and in vivo micronucleus test were performed in compliance with the Organisation for Economic Cooperation and Development (OECD) guidelines for the testing of chemicals under the modern Good Laboratory Practice Regulations [5]. The results of this investigation will provide additional information relevant to the safety evaluation of titanium dioxide, GST exposure.
Materials and MethodsBacterial reverse mutation testThe bacterial reverse mutation test was conducted in accordance with the OECD guideline No. 471 for the testing of chemicals [6]. The tester strains purchased from Molecular Toxicology Inc. (Boone, NC, USA) used in this study were Salmonella typhimurium TA98, TA100, TA1535, and TA1537, and Escherichia coli WP2uvrA in the absence and presence of metabolic activation system. The metabolic activation system was prepared by mixing S9 metabolic activation (Molecular Toxicology Inc.) with Cofactor 1 from Wako Pure Chemical Industries Ltd (Osaka, Japan), giving a final concentration of 5% (volume/volume) S9. The tester strains were cultured in 2.5% nutrient broth No. 2 (Oxoid Ltd, Basingstoke, UK) in a 37 °C shaking incubator (120 rpm) for approximately 10 h. The mutagenicity test was performed by mixing test substance and tester strains, which was cultured overnight in the presence and absence of the S9 mixture. Next, the mixture was incubated in a water bath for 20 min at 37 °C, mixed with top agar and a minimal amount of histidine-biotin (for S. typhimurium strains) or tryptophan (for E. coli strain), and then poured onto the surface of a gamma-ray sterile Falcon® Petri dish (Thermo Fisher Scientific, Waltham, MA, USA) containing about 15 mL of minimal glucose agar. The finished plates were incubated for 72 h at 37 °C. The number of revertant colonies was then counted. Three plates per treatment group were conducted, and the results were tabulated as the mean ± standard deviation for each condition.
In vitro chromosomal aberration testThe clastogenicity of GST was evaluated for its ability to induce chromosomal aberrations in Chinese hamster lung (CHL) fibroblast cells. The chromosomal aberration test was conducted in accordance with the OECD guideline No. 473 for the testing of chemicals [7]. A clonal subline of CHL cells was obtained from the American Type Culture Collection (Rockville, MD, USA). The karyotype of the CHL cells consisted of 25 chromosomes. The CHL cells were grown in Minimum Essential Eagle’s Medium, supplemented with 10% fetal bovine serum (FBS), 50 U/mL penicillin, and 50 μg/mL streptomycin (Gibco BRL Life Technologies Inc., Gaithersburg, MD, USA) at 37 °C in a humidified atmosphere containing 5% CO2. Short-time treatment method (with or without metabolic activation system) and continuous treatment method were conducted. Mitomycin C (Sigma-Aldrich Co., St Louis, MO, USA) was used as a positive control, both with and without the S9 mixture. After 22 h of incubation, colcemid was added to the cultures at a final concentration of 0.2 μg/mL, and meta phase cells were harvested by trypsinization and centrifugation. The cells were swelled by adding hypotonic (0.075 M) KCl solution for 20 min at 37 °C, and then washed two times in ice-cold fixative (ethanol to glacial acetic acid, 3:1). A few drops of cell pellet suspension were dropped onto precleaned glass microscope slides and air-dried. The slides were stained with 5% Giemsa buffer solution (Thermo Fisher Scientific, Waltham, MA, USA). The number of cells with chromosomal aberrations was recorded on 300 well spread metaphases. Aberration frequencies, defined as aberrations observed, were divided by the number of cells counted, and were analyzed using Fisher’s exact test with Dunnett’s adjustment.
In vivo micronucleus testThe micronucleus test was performed in accordance with the OECD test guideline No. 474 for the testing of chemicals [8]. The 7-week-old ICR mice were randomized into groups containing five mice each. As a result of the vehicle test, the test substance was suspended in sterile distilled water at 200 mg/mL, and there was no heat, foaming, or discoloration when mixed, so sterile distilled water was selected as the vehicle. The test substance was administered orally in three doses in volumes of 10 mL/kg. It was given twice with a 24 h interval in between, and test subjects were sacrificed by CO2 inhalation. The bone marrow was flushed with FBS (Gibco BRL Life Technologies Inc.). The centrifuge tubes were selected for centrifugation of the cells and centrifuged at 1000 rpm for 5 min. The suspended cells were smeared on a clean slide. The smeared slides were air-dried and thereafter fixed for 5 min in methanol. The 40 μg/mL acridine orange was dropped on the methanol-fixed slides for the observation.
In scoring the preparations, micronuclei were counted in polychromatic erythrocytes (PCE) and separately in monochromatic erythrocytes. The rate of micronucleated cells, expressed as a percentage, was based on the total of PCE present in the scored optic fields. The scoring of micronucleated normocytes was used to recognize the presence of artifacts (which is rare in mouse preparations), which provided additional interesting information on the mode of action of the test substances. Generally, an incidence of more than one micronucleated normocyte per 1000 PCE indicates an effect on cell stages, especially post-S-phase.
Test procedureProperties of GSTThe characterization of GST is shown in Figure 1, 2 and 3, and individually compared to another titianium dioxide, P-25. The zeta potential is the potential between droplet surface and dispersing liquid medium and can be used to estimate surface charge of the droplets in the dispersion medium. Also, it was well known as indicator of the droplet stability, where values more positive than +30 mV and more negative than -30 mV indicate good stability against coalescence [9]. The estimated value for GST showed that GST has a negative value (-35.4 mV) thought to be good stability and it was thought to be a property to be less agglomeration nature (Figure 1). The particle size and distribution (95.8 ± 46.3 nm, 46 – 270 nm) showed that GST was considered to have materials of various sizes and was difficult to be classified as a nanomaterial considering the definition of nanomaterials (< 100 nm) (Figure 2–3).
Animal husbandary and maintenanceThe 6-week-old male ICR mice were obtained from the Orient Bio Inc. (Gyeonggi, Korea). The animals were used after 7 days of acclimatization and housed animal to a stainless wire cage in a room with barrier system controlled for the light-dark cycle (12–12 h), air exchange (10–20 changes/h), temperature (19–25 °C) and relative humidity (30–70%) during the study. The animals were fed with Rodent Diet 20 5053 (LabDiet, USA) and provided with the reverse osmosis water ad libitum. The animals were maintained in accordance with the Guide for the Care and Use of Laboratory Animals [10].
Clinical signs and Body weightDuring the study, all animals were observed once daily after treatment for any clinical signs of toxicity and mortality. The body weight of each mouse was measured at the time of animal receipt, grouping, before administration and autopsy.
Statistical analysisThe result of the statistical evaluation was deemed to be statistically significant when the P-value was less than 0.05. We used the Kruskal–Wallis H test and Dunnett’s test for differences in numbers of micronucleated polychromatic erythrocytes (MNPCE) between the treated and negative control groups; the Mann–Whitney U test for differences in numbers of MNPCE between the positive and negative control groups; analysis of variance and Dunnett’s test for differences in the PCE/(PCE + normochromatic erythrocyte (NCE)) ratio between the treated and negative control groups; the Student’s t-test for differences in the PCE/(PCE + NCE) ratio between the positive and negative control groups; and analysis of variance and Dunnett’s test for comparison of animal body weight at the time of euthanasia.
Results and DiscussionBacterial reverse mutation testIn the concentration range-finding test (data not shown), it was found to have a nontoxic effect in all tester strains of S. typhimurium (TA98, TA100, TA1535, and TA1537) and in E. coli WP2uvrA at a dose of 5000 μg per plate in the presence and absence of S9 mixture. Based on the data from the concentration range-finding test, we selected 5000 μg per plate as the highest dose. As shown in Table 1, none of the tester strains showed any increase in the number of revertant colonies in comparison with the negative control group when the bacteria were treated with GST at 312.5, 625, 1250, 2500, and 5000 μg per plate, regardless of metabolic activation. On the other hand, the positive control group used in the assays with the presence or absence of S9 mixture showed positive responses by the respective test strains, as evidenced by the number of revertant colonies being greater than 2-fold of the respective those of negative control group. The bacterial reverse mutation test detects point mutations, which caused many human genetic diseases and play an important role in tumor initiation and development [11]. This assay is a widely accepted short-term test to identify substances that can produce genetic damage leading to gene mutations [12]. In the present study, we found no positive mutagenic responses to GST in any of the tester strains compared with the negative control both with and without the S9 mixture.
In vitro chromosomal aberration testInitially, a concentration range-finding test was performed to determine the test doses for use in the in vitro chromosome aberration test. The highest concentration was determined to be 50 μg/mL in a short-term treatment assay in the absence of S9 mixture (referred to as –S9 mix) and to be 80 μg/mL in the presence of S9 mixture (referred to as +S9 mix), to be 30 μg/mL continuous treatment test (referred to as 24 h exposure). The in vitro chromosome aberration test was conducted with GST at concentrations of 0, 12.5, 25 and 50 μg/mL without metabolic activation. In the presence of S9 mixture, the concentrations were 0, 20, 40 and 80 μg/mL. Except for the positive control group, cells arrested in metaphase with structural aberrations were less than 5% (Table 2). Chromosome aberrations are the classical genotoxic response to tumor initiation and development processes [13]. The purpose of the in vitro chromosome aberration test is to identify agents that cause structural chromosome aberrations in cultured mammalian cells [14]. Our results indicated no significant increase in the number of metaphases with structural aberrations at the four concentrations in GST, regardless of metabolic activation. The results of the in vitro chromosomal aberration test suggest that GST was not considered to have the ability to induce the chromosomal aberrations in CHL/IU cells under the present experimental conditions.
In vivo micronucleus testBased on the results of a dose range-finding test (data not shown), the highest dose of test substance in main test was determined at 2000 mg/kg body weight. The PCE/(PCE + NCE) ratios were used as an index of bone marrow cytotoxicity. The ratios did not show any significant difference in the GST treatment groups in comparison with the negative control group. The MNPCE frequencies were not statistically significant and did not show any dose-dependent pattern among the three treatment groups in comparison with the negative control group. On the other hand, the positive control group showed a significantly increased MNPCE frequency in comparison with the negative control group and GST treatment groups (Table 3). In all groups of main test, there was no dead animal during the study period and no significant difference in body weights compared with the negative control group (Table 4). The micronucleus test (MNT) is a useful assay for the detection of mutagenic substances, thus altering the equitable distribution of chromosomes during cell division [15]. The present micronucleus test was performed using the bone marrow cells of specific pathogen-free male ICR mice. GST did not induce any significant increases in MNPCEs, and there was no significant decrease in the PCE/(PCE+NCE) ratio up to 2000 mg/kg body weight in the GST treatment groups compared with the negative control group. From these results, GST was determined not to induce an increased frequency of micronuclei in the bone marrow cells of male ICR mice under the present experimental condition. Also, GST had no cytotoxicity effects in the bone marrow cells.
ConclusionsTitanium dioxide (TiO2) is a natural oxide of the elements titanium with low toxicity, and negligible biological effects. In spite of the extensively use of TiO2, the biological effects are still not completely elucidated [16]. To determine the safety of TiO2 in the human health, systemic toxicological studies must be performed using various experimental models to predict the potential toxicity. Genotoxicity tests have been used mainly for the prediction of in vivo genotoxicity and carcinogenicity of chemicals, because compounds that show positive results in these tests have carcinogenic and/or mutagenic potential in humans [17]. In this study, the potential genotoxicity of the GST was examined in the three battery of in vitro and in vivo genotoxicity tests (bacterial reverse mutation test, chromosome aberration teat, and bone marrow micronucleus test) in accordance with the OECD Test Guidelines and Principles of Good Laboratory Practice.
The bacterial reverse mutation test detects point mutations, which caused many human genetic diseases and play an important role in tumor initiation and development [11, 18]. This test is a widely accepted short-term test to identify substances that can produce genetic damage leading to gene mutations [12, 19]. In the present study, we found no positive mutagenic responses to GST in any of the tester strains compared with the negative control, both with and without the S9 mixture.
Chromosome aberrations are the classical genotoxic responses to tumor initiation and development processes [13]. The purpose of the in vitro chromosome aberration test is to identify agents that cause numerical and structural chromosome aberrations in cultured mammalian cells [14]. Our results showed no significant increase in the number of metaphases with numerical and structural chromosome aberrations at the three concentrations of GST tested, regardless of metabolic activation. Based on the results of the in vitro chromosome aberration test, GST was not considered to have the ability to induce chromosomal aberrations in CHL cells under the present experimental conditions.
The micronucleus test detects mutagenic substances that alter the equitable distribution of chromosomes during cell division [15, 20]. The present micronucleus test was performed using the bone marrow cells of specific pathogen-free male ICR mice. GST did not induce any significant increases in MNPCEs, and there was no significant decrease in the PCE/(PCE+NCE) ratio up to 2000 mg/kg body weight in the GST treatment groups compared to that of the negative control group. From these results, GST was determined not to induce an increased frequency of micronuclei in the bone marrow cells of male ICR mice under the present experimental conditions. In addition, GST showed no cytotoxic effects in the mouse bone marrow cells.
TiO2 material, which is covered in this paper, GST (100% anatase) was prepared from the precipitated sludge using TiCl4 used as a coagulant to remove total phosphorus in the wastewater. The experimental genotoxicity data were restricted to studies from the largely applied comet assays, which is not validated by the OECD yet. In vivo data are also available for TiO2 materials, even if in vivo data are considered of higher relevance than in vitro data to conclude on genotoxicity, they are quite limited.
In conclusion, GST was regarded to have no genotoxic or cytotoxic potential in both in vitro and in vivo system under our current experimental condition. These results suggest that GST is relatively safe on the mutagenicity and genotoxicity, and are expected to provide some information on the risk assessment process.
AcknowledgementThis work was supported by a grant (19SCIP-B145906-02) from the Korea Agency for Infrastructure Technology Advancement (KAIA) by Ministry of Land, Infrastructure and Transport of Korea government (MOLIT), Republic of Korea.
NotesCRediT author statement
JSK: Conceptualization, Methodology, Writing-Original draft preparation, MHJ: Conceptualization, Methodology, WritingOriginal draft preparation, HSS: Supervision, Writing-Reviewing and Editing, MKP: Supervision, HJP: Resources, SSN: Project administration, Writing-Reviewing and Editing
References1. Emerich DF, Thanos CG. Nanotechnology and medicine. Expert Opin Biol Ther 2003;3(4):655-63 https://doi.org/10.1517/14712598.3.4.655.
2. Orlov A, Chan MS, Jefferson DA, Zhou D, Lynch RJ, Lambert RM. Photocatalytic degradation of water-soluble organic pollutants on TiO2 modified with gold nanoparticles. Environ Technol 2006;27(7):747-52 https://doi.org/10.1080/09593332708618685.
3. Oberdörster G, Maynard A, Donaldson K, Castranova V, Fitzpatrick J, Ausman K, et al. Principles for characterizing the potential human health effects from exposure to nanomaterials: elements of a screening strategy. https://doi.org/10.1186/1743-8977-2-8.
4. Chen T, Yan J, Li Y. Genotoxicity of titanium dioxide nanoparticles. J Food Drug Anal 2014;22(1):95-104 https://doi.org/10.1016/j.jfda.2014.01.008.
5. Organization for Economic Co-operation and Development (OECD). OECD Series on principle of good laboratory practice and compliance monitoring, No.1 (Revised): OECD Principles of Good Laboratory Practice, ENV/MC/CHEM(98)17. [cited Dec 1, 2021]. Available from: https://one.oecd.org/document/ENV/MC/CHEM(98)17/en/pdf.
6. Organization for Economic Co-operation and Development (OECD). OECD Guidelines for the testing of chemicals, Section 4, Test No. 471. [cited Dec 1, 2021]. Available from: https://www.oecd-ilibrary.org/environment/oecd-guidelinesfor-the-testing-of-chemicals-section-4-health-effects_20745788?page=3.
7. Organization for Economic Co-operation and Development (OECD). OECD Guidelines for the testing of chemicals, Section 4, Test No. 473. [cited Dec 1, 2021]. Available from: https://www.oecd-ilibrary.org/environment/oecd-guidelinesfor-the-testing-of-chemicals-section-4-health-effects_20745788?page=3.
8. Organization for Economic Co-operation and Development (OECD). OECD Guidelines for the testing of chemicals, Section 4, Test No. 474. [cited Dec 1, 2021]. Available from: https://www.oecd-ilibrary.org/environment/oecd-guidelinesfor-the-testing-of-chemicals-section-4-health-effects_20745788?page=3.
9. Alexandru Mihai Grumezescu. Self-nanoemulsifyomg drug delivery systems (SNEDDS) and self-microemulsifying drug delivery systems (SMEDDS) as lipid nanocarriers for improving dissolution rate and bioavailability of poorly solutble drugs. In: Lipid Nanocarriers for drug targeting. Elsevier Science; 2018. 473-508.
10. Clark JD, Gebhart GF, Gondor JC, Keeling ME, Kohn DF. The 1996 guide for the care and use of laboratory animals. ILAR J 1997;38(1):41-48 https://doi.org/10.1093/ilar.38.1.41.
11. Zeiger E. Bacterial mutation assays. Methods Mol Biol 2013;1044: 3-26 https://doi.org/10.1007/978-1-62703-529-3_1.
12. Mortelmans K, Zeiger E. The Ames Salmonella/microsome mutagenicity assay. Mutat Res 2000;455(1-2):29-60 https://doi.org/10.1016/s0027-5107(00)00064-6.
13. Chen Q, Tang S, Jin X, Zou J, Chen K, Zhang T, et al. Investigation of the genotoxicity of quinocetone, carbadox and olaquindox in vitro using Vero cells. Food Chem Toxicol 2009;47(2):328-334 https://doi.org/10.1016/j.fct.2008.11.020.
14. Ishidate M, Jr, Sofuni T. The in vitro chromosomal aberration test using Chinese Hamster Lung (CHL) fibroblast cells in culture. In: Ashby J, editor. Progress in mutation research. Vol 5. Elsevier Science; 1985. 427–432.
15. Kirsch-Volders M, Sofuni T, Aardema M, Albertini S, Eastmond D, Fenech M, et al. Report from the in vitro micronucleus assay working group. Mutat Res 2003;540(2):153-163 https://doi.org/10.1016/j.mrgentox.2003.07.005.
16. Grande F, Tucci P. Titanium dioxide nanoparticles: a risk for human health. Mini Rev Med Chem 2016;16(9):762-769 https://doi.org/10.2174/1389557516666160321114341.
17. Rama RP, Kaul CL, Jena GB. Genotoxicity testing, a regulatory requirement for drug discovery and development: impact of ICH guidelines. Indian Journal of Pharmacology 2002;34(2):86-99.
18. Park CG, Cho HK, Shin HJ, Park KH, Lim HB. Comparison of mutagenic activities of various ultra-fine particles. Toxicol Res 2018;34(2):163-172 https://doi.org/10.5487/TR.2018.34.2.163.
19. Shin HJ, Cho HG, Park CK, Park KH, Lim HB. Comparative in vitro biological toxicity of four kinds of air pollution particles. Toxicol Res 2017;33(4):305-313 https://doi.org/10.5487/TR.2017.33.4.305.
20. Kim JY, Ri Y, Do SG, Lee YC, Park SJ. Evaluation of the genotoxicity of ginseng leaf extract UG0712. Lab Anim Res 2014;30(3):104-111 https://doi.org/10.5625/lar.2014.30.3.104.
Figure 1.Characterization of TiO2 particles (GST) compared to P-25 analyzed by Korea TECH: (a) negative zeta potential (-35.4 mV, 30 mg/mL); (b) size distribution by intensity (mean: 336.8 nm); (c) positive zeta potential (32.3 mV, 30 mg/mL); (d) size distribution by intensity (mean: 302.2 nm). 
Figure 2.Characterization of TiO2 particles (GST): SEM (scanning electron microscope) image analyzed by KRICT. 
Figure 3.Characterization of TiO2 particles (GST); (a) A particles dispersed in 99.9 % EtOH was deposited on a copper grid and analyzed using TEM (Transmission electron microscope) image by Korea Basic Science Institute, (b) Size distribution (95.8 ± 46.3 nm, 46 – 270 nm,) of the imaged GST(image J software). 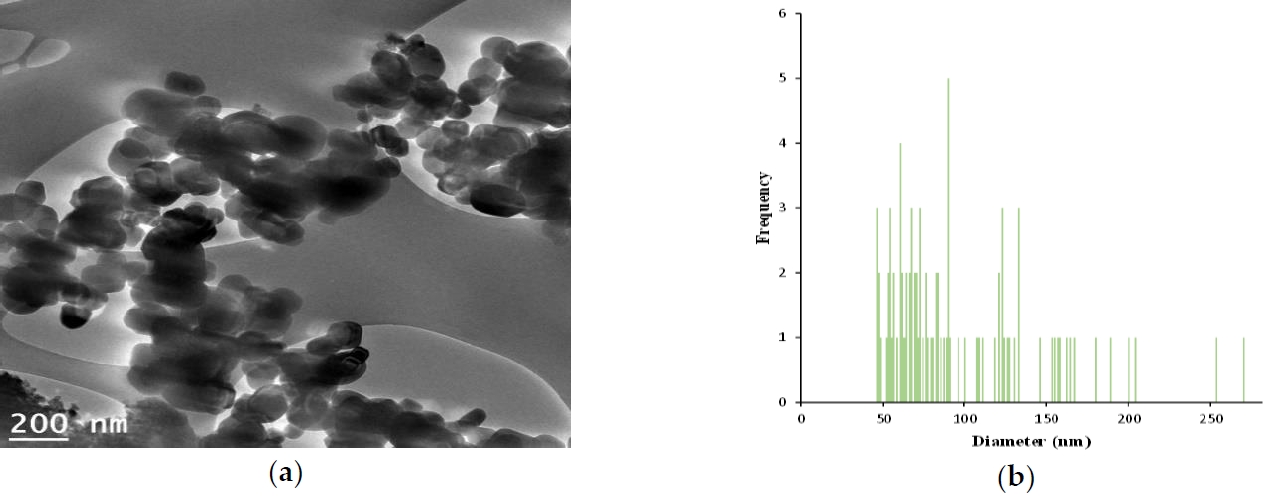
Table 1.Numbers of revertant colonies induced by GST in Salmonella typhimurium (TA98, TA100, TA1535, TA1537) and Escherichia coli (WP2uvrA) with and without metabolic activation (S9 mixture).
Table 2.Chromosome analysis of GST in Chinese hamster lung fibroblast cells with and without metabolic activation (S9 mixture)
Table 3.Frequencies of MNPCE per 2,000 PCE in the bone marrow of ICR mice exposed to GST
Table 4.Body weights of ICR mice exposed to GST |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||