AbstractThe coronavirus disease of 2019 (COVID-19) has become a global pandemic with rapid rate of transmission and fatalities worldwide. Scientists have been investigating a host of drugs that may be rechanneled to fight this malaise. Thus, in this current computational study we carried out molecular docking experiments to assess the bridging potentials of some commercial drugs such as chloroquine, hydroxychloroquine, lopinavir, ritonavir, nafamostat, camostat, famotidine, umifenovir, nitazoxanide, ivermectin, and fluvoxamine at the interface between human ACE2 and the coronavirus spike glycoprotein complex. This is aimed at ascertaining the ability of these drugs to bridge and prevent the complexing of these two proteins. The crystal structure of human ACE2 and the coronavirus spike glycoprotein complex was retrieved from protein database, while the selected drugs were retrieved from PubChem data base. The proteins and drugs were prepared for docking using Cresset Flare software. The docking was completed via AutoDock Vina module in Python Prescription software. The best hit drugs with each receptor were selected and their molecular interactions were analyzed using BIOVIA’s Discovery Studio 2020. The best hit compounds on the human ACE2 were the lopinavir (-10.1 kcal/mol), ritonavir (-8.9 kcal/mol), and nafamostat (-8.7 kcal/mol). Ivermectin, nafamostat, and camostat with binding energy values -9.0 kcal/mol, -7.8 kcal/mol, and -7.4 kcal/mol respectively were the hit drugs on the coronavirus spike glycoprotein. Nafamostat showed a dual bridging potential against ACE2 and spike glycoprotein, and could therefore be a promising lead compound in the prevention and control of this disease.
IntroductionThe coronavirus disease of 2019 (COVID-19) has become a global pandemic with rapid rate of transmission and high levels of fatalities in many countries [1,2]. The numbers of deaths from this disease have surpassed 3.7 million worldwide as at June, 2021 [3]. Various endeavors have been made to design novel inhibitors or utilize drug repurposing approach to recognize anti-COVID-19 drugs, which can go about as promising inhibitors of COVID-19 proteins [4]. The spike protein of the coronavirus is found on the surface of the virus and has huge protrusions or peplomers which appear like crowns hence the name CORONA (the Latin word for crown). It has three segments namely: the big ectodomain, the transmembrane domain and the intracellular tail. The receptor-binding subunits S1 and S2 are located in the ectodomain area. At infection, the S1 receptor-binding subunit binds to the host receptor while the S2 receptor-binding subunit fuses with the host, releasing the genome of the virus into the cell [5]. The angiotensin-converting enzyme 2 (ACE2) was identified two decades ago and received its name due to its homology with ACE [6]. The ACE2 is a type I transmembrane glycoprotein which is considered a critical receptor for coronavirus infections as well as a therapeutic target for COVID-19 patients. The virus attaches to the host ACE2 receptor and then uses the spike protein to enter target cells [7].
Numerous studies related to identifying effective therapeutics against COVID-19 have been reported [8,9]. Sadly, there are no particular treatments endorsed by perceived globally recognized health organizations, like the WHO and US Food and Drug Administration (FDA). Scientists have been investigating a plethora of drugs that may be repurposed either alone or in combination with other known drugs to fight COVID-19 [4,10]. Currently, fifteen drugs including chloroquine, hydroxychloroquine, lopinavir, ritonavir, nafamostat, camostat, famotidine, umifenovir, nitazoxanide, ivermectin and fluvoxamine are being studied with the hope of discovering a treatment for this disease [11]. Regardless of their accomplishment in clinical reports, none have acquired endorsement for use as their mechanisms of action on the virus are yet to be elucidated.
In this study, we performed an in silico investigation of the bridging potentials of some of the aforementioned drug candidates at the interface between human ACE2 and the coronavirus spike glycoprotein complex. The binding affinity of these drug compounds on the targets would be used to assess their abilities to block the complexing of the two proteins.
Materials and MethodsProtein receptors and ligand retrieval and preparationsThree dimensional (3D) structures of the drug compounds were retrieved from PubChem web server in simple document format (SDF). They were optimized using Open babel in Python Prescription (version 0.8) which converted the ligands energetically to the most stable structures using Merk Molecular Force Field 94 (MMFF94). Similarly, the 3D X-ray crystallographic structure of the human ACE2 and SARS-CoV-2 spike glycoprotein (Figures 1 and 2) was retrieved from the protein data bank (PDB) with ID 6LZG [12]. The proteins were then prepared for docking and minimized using the relevant tools in Cresset Flare© software, version 4.0 [13]. The protein minimization was based on the General Amber Force Field (GAFF), with gradient cutoff of 0.200 kcal/mol/A and iterations was set to 2000 iterations [8,14].
Molecular docking procedureThe molecular docking was achieved through flexible docking protocol [8,15]. In brief; Python Prescription 0.8, a suite housing Auto Dock Vina module, was employed for the molecular docking study of the selected compounds with both protein receptors. The specific target site on each receptor where complexing occurred between ACE2 and spike glycoprotein [12] were set using grid box dimensions 17.9656 × 43.3347 × 22.1744 Å and 21.9505 × 42.7540 × 41.9980 Å for ACE2 and spike glycoprotein respectively. Also, the centres for both receptors were adjusted based on their active site. At the end of the molecular docking, ten binding poses of the protein-ligand complex was generated and scoring result was created after retrieving the text file for comparative analysis. BIOVIA’s Discovery Studio 2020 was deployed for the molecular interaction analyses both in 2D and 3D presentation.
Results and DiscussionThe binding affinities of these drugs on the human ACE2 and spike glycoprotein targets are shown in (Table 1).
The compounds with the highest binding affinities on the human ACE2 were the antiretroviral drugs lopinavir (-10.1 kcal/mol), ritonavir (-8.9 kcal/mol), and the anti-inflammatory drug nafamostat (-8.7 kcal/mol). The interactions of these drugs with the amino acids at the target site of ACE2 protein are shown in (Figure 3).
The amino acids on the ACE2 target that interacted with the drugs and the nature of the interactions between them are summarized in (Table 2).
Similar amino acids interacted withlopinavir and ritonavir on the human ACE2 protein. Pi-Pi stacked and Pi-Alkyl interactions occurred at PHE40 and ARG393 of this protein for both drugs, while TRP69 formed a hydrogen bond with Ritonavir and a Pi-Pi T-shaped bond with Lopinavir. Nafamostat interacted with ARG393 via hydrogen bonding and Pi-Alkyl interactions and with PHE40 by Pi-Pi T-shaped interaction.
Ivermectin, nafamostat, and camostat had the highest binding affinity on the spike glycoprotein with binding free energy values -9.0 kcal/mol, -7.8 kcal/mol, and -7.4 kcal/mol respectively. This result indicated that the binding of these drugs at the spike glycoprotein target could inhibit its penetration into the human ACE2. The interactions of these drugs with the amino acids at this site are shown in (Figure 4).
The amino acids on the spike glycoprotein target that interacted with these drugs and the nature of the interactions between them are summarized in (Table 3).
Hydrogen bonds were prevalent in the binding of all the drugs with the spike glycoprotein. The binding interaction of ivermectin with the amino acid residues at the target site was formed by three hydrogen bonds. Nafamostat and camostat were bound to the residues by four hydrogen bonds together with other interactions. Hydrogen bonds are strong interactions that could confer high stability to the drugs at protein targets. The amino acid residues that interacted with these drugs were markedly different and suggested that their inhibition of the action of the spike glycoprotein followed different mechanisms.
DiscussionChloroquine, hydroxychloroquine, lopinavir, ritonavir, nafamostat, camostat, famotidine, umifenovir, nitazoxanide, ivermectin, and fluvoxamine were used for the in silico binding studies. Chloroquine is a well-known anti-malarial drug for the treatment of malarial caused by Plasmodium vivax, Plasmodium ovale, Plasmodium falciparium, and Plasmodium malariae. Hydroxychloroquine is used to prevent and treat malaria in areas where malaria remains sensitive to chloroquine. Other uses include treatment of rheumatoid arthritis, lupus, and Porphyria cutaneatarda. It is taken orally often in the form of hydroxychloroquine sulfate [16,17] Lopinavir and ritonavir are HIV protease inhibitors used as a combination product with other HIV medications to control HIV infections. Ritonavir acts as a pharmacokinetic booster to other protease inhibitor to give a highly active antiretroviral effect [18] Nitazoxanide is used in the treatment of diarrhea caused by Cryptosporidium parvum or Giardia lamblia [19]. Nafamostat, a broad-spectrum protease inhibitor is used as an anticoagulant in patients with hemorrhagic cases. It is also anti-inflammatory and increases mucus clearance in the airways. It prevents blood clot formation during extracorporeal circulation in patients undergoing continuous renal replacement therapy and extra corporeal membrane oxygenation. Both Nafamostat and Camostat were developed in Japan as treatments for pancreatitis and some other diseases. They can be used as lone drugs or in combination with other antiviral drugs [20]. Famotidine is used to treat and prevent intestinal and gastric ulcers, heartburn, esophagitis, gastroesophageal reflux disease as well as Zollinger-Ellison syndrome. It decreases production of acid in the stomach and relieves cough [21]. Umifenovir, licensed in China and Russia is an antiviral medication for the treatment of influenza and other respiratory infections. It is also indicated against other viruses, enveloped or not, that are responsible for infectious diseases such as hepatitis B and C, gastroenteritis, hemorrhagic fevers or encephalitis [22]. Fluvoxamine is an antidepressant used to treat obsessive-compulsive disorder and also restores the balance of serotonin in the brain [23]. Ivermectin is an antihelmintic used against parasitic infestations. It is used for the treatment of onchocerciasis, strongyloidiasis, ascariasis, filariases, mites, lice, and scabies. In veterinary medicine, it is used to prevent and treat heartworm and acariasis, among other indications [24].
The binding free energy values displayed by lopinavir, ritonavir, and nafamostat after the docking study (Table 1) indicated that these compounds could disrupt the complexing of the spike glycoprotein with the human ACE2 [25]. Similarly, the interactions of lopinavir and ritonavir with the amino acids of human ACE2 (Figure 3) suggested that the mechanism of action of these drugs are similar [26]. Three hydrogen bond interactions between nafamostat and ACE2 protein conferred the drug with greater stability at the protein target [27]. It is of interest to note that two amino acid residues PHE40 and ARG393 (Table 2) are found to be important sites on the ACE2 protein for the interaction and action of these drugs. On the other hand, Ivermectin, nafamostat, and camostat had the highest binding affinity on the spike glycoprotein (Table 2) which indicated that the binding of these drugs at the spike glycoprotein target could inhibit its penetration into the host’s cell via the human ACE2. Furthermore, the interactions of these drugs with the amino acids at the site of contact in the spike glycoprotein with the human ACE2 were mostly by hydrogen bonding (Figure 4 and Table 3). The hydrogen interactions reported herein, are typically strong interactions that could confer high stability to the drugs at protein targets. Different amino acid residues however were found to interact with these drugs and suggested that their inhibition of the activity of the spike glycoprotein followed varying mechanisms.
ConclusionsThe potentials of chloroquine, hydroxychloroquine, lopinavir, ritonavir, nafamostat, camostat, famotidine, umifenovir, nitazoxanide, ivermectin, and fluvoxamine to inhibit the interaction of the spike glycoprotein of the coronavirus and the human ACE2 protein were studied in silico. Molecular docking was performed on the zone harboring the amino acids involved in the complexing of these two proteins. Lopinavir, ritonavir, and nafamostat showed good binding affinity on ACE2, while ivermectin, nafamostat, and camostat had the best binding affinity on the coronavirus spike glycoprotein. The good binding affinity demonstrated by nafamostat at both ACE2 and spike glycoprotein showed that this drug could impact dual action in blocking the interactions of the two proteins and hence infection by COVID-19.
RecommendationThe unabated fatality of COVID-19 all over the world calls for urgent attention in finding an effective and affordable cure for this disease. The drug nafamostat has shown good binding affinity on the human ACE2 and spike glycoprotein of the coronavirus. This drug should therefore be subjected to further screening to establish its efficacy and possible implementation in controlling this worldwide pandemic.
ACKNOWLEDGEMENTSThe authors are grateful to ChemSolvers Research and Computational Laboratory Studio, Owerri, Nigeria, for assisting in the in silico studies.
Conflict of interestThe authors declare no conflict of interest regarding the publication of this manuscript.
NotesCRediT author statement
CED: Conceptualization, Data curation, Supervision, Methodology, Software HIU: Conceptualization, Supervision, Methodology, Data curation, Software. IAD: Visualization, Investigation. UEE: Visualization, Investigation. LCN: Original draft preparation, Writing- Reviewing and Editing. CEE: Original draft preparation, Writing- Reviewing and Editing.
References1. Enyoh CE, Verla AW, Verla EN. Novel coronavirus (SARS-CoV-2) and airborne microplastics. Journal of Materials and Environment Science 2020;11(9):1454-1461 https://doi.org/10.5281/zenodo.3738452.
2. Robson B. Computers and viral diseases. Preliminary bioinformatics studies on the design of a synthetic vaccine and a preventative peptidomimetic antagonist against the SARS-CoV-2 (2019-nCoV, COVID-19) coronavirus. Computers in Biology and Medicine 2020;119: 103670 https://doi.org/10.1016/j.compbiomed.2020.103670.
3. Worldometer (2021). COVID-19 coronavirus pandemic. Assessed on June 7, 2021. www.worldometers.info/coronavirus/.
4. Khan RJ, Jha RK, Amera GM, Jain M, Singh E, Pathak A, Singh RP, Muthukumaran J, Singh AK, et al. Targeting SARS-CoV2: A systematic drug repurposing approach to identify promising inhibitors against 3C-like proteinase and 20 -O-ribose methyltransferase. Journal of Biomolecular Structure and Dynamics 2020;1-14 https://doi.org/10.1080/07391102.2020.1753577.
5. Chan JFW, Yuan S, Kok KH, To KKW, Chu H, Yang J, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet 2020;395: 514-523 https://doi.org/10.1016/S0140-6736(20)30154-9.
6. Peiris JSM, Guan Y, Yuen KY. Severe acute respiratory syndrome. Nature Medicine 2004;10: 88-97 https://doi.org/10.1038/nm1143.
7. Cheng H, Wang Y, Wang GQ. Organ-protective effect of angiotensin-converting enzyme 2 and its effect on the prognosis of COVID-19. Journal Medical Virol 2020;92: 726-730 https://doi.org/10.1002/jmv.25785.
8. Umar HI, Josiah SS, Saliu TP, Jimoh TO, Ajayi A, Danjuma JB. In-silico analysis of the inhibition of the SARS-CoV-2 main protease by some active compounds from select African plants. J Taibah Univ Medical Sci 2021;16(2):162-176 https://doi.org/10.1016/j.jtumed.2020.12.005.
9. Duru CE, Duru IA, Adegboyega AE. In silico identification of compounds from Nigella sativa seed oil as potential inhibitors of SARS-CoV-2 targets. Bull Natl Res Cent 2021;45(1):1-13 https://doi.org/10.1186/s42269-021-00517-x.
10. Muralidharan , Nisha , Sakthivel R, Velmurugan D, Gromiha MM. Computational studies of drug repurposing and synergism of lopinavir, oseltamivir and ritonavir binding with SARS-CoV-2 protease against COVID-19. J Biomol Struct Dyn 2020;1-6 https://doi.org/10.1080/07391102.2020.1752802.
11. Leah S. 15 drugs being tested to treat COVID-19 and how they would work. Nature Medicine 2020;https://doi.org/10.1038/d41591-020-00019-9.
12. Wang Q, Zhang Y, Wu L, Niu S, Song C, Zhang Z, et al. Structural and functional basis of SARS-CoV-2 entry by using human ACE2. Cell 2020;181(4):894-904 https://doi.org/10.1016/j.cell.2020.03.045.
13. Cresset. Flare software. version 4.0. 2020. Assessed on May 21, 2021.https://www.cresset-group.com/software/.
14. Stroganov OV, Novikov FN, Zeifman AA, Stroylov VS, Chilov GG. TSAR, a new graph-theoretical approach to computational modeling of protein side-chain flexibility: Modeling of ionization properties of proteins. Proteins 2011;79(9):2693-2710 https://doi.org/10.1002/prot.23099.
15. Trott O, Olson AJ. AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem 2010;319(2):455-461 https://doi.org/10.1002/jcc.21334.
16. Al-Bari MAA. Chloroquine analogues in drug discovery: new directions of uses, mechanisms of actions and toxic manifestations from malaria to multifarious diseases. J Antimicrob Chemother 2015;70(6):1608-1621 https://doi.org/10.1093/jac/dkv018.
17. Ben-Zvi I, Kivity S, Langevitz P, Shoenfeld Y. Hydroxychloroquine: from malaria to autoimmunity. Clin Rev Allergy Immunol 2012;42(2):145-153 https://doi.org/10.1007/s12016-010-8243-x.
18. Doggrell SA. Does lopinavir measure up in the treatment of COVID-19? Expert Opin Investig Drugs 2020;29(8):793-796 https://doi.org/10.1080/13543784.2020.1777277.
19. Rossignol JF, Lopez-Chegne N, Julcamoro LM, Carrion ME, Bardin MC. Nitazoxanide for the empiric treatment of pediatric infectious diarrhea. Trans R Soc Trop Med Hyg 2012;106(3):167-173 https://doi.org/10.1016/j.trstmh.2011.11.007.
20. Yamamoto M, Kiso M, Sakai-Tagawa Y, Iwatsuki-Horimoto K, Imai M, Takeda M, Kinoshita N, et al. The anticoagulant nafamostat potently inhibits SARS-CoV-2 S protein-mediated fusion in a cell fusion assay system and viral infection in vitro in a cell-type-dependent manner. Viruses 2020;12(6):629 https://doi.org/10.3390/v12060629.
21. Wesdorp IC, Dekker W, Festen HP. Efficacy of famotidine 20 mg twice a day versus 40 mg twice a day in the treatment of erosive or ulcerative reflux esophagitis. Dig Dis Sci 1993;38(12):2287-2293 https://doi.org/10.1007/BF01299910.
22. Blaising J, Polyak SJ, Pécheur EI. Arbidol as a broad-spectrum antiviral: an update. Antiviral Res 2014;107: 84-94 https://doi.org/10.1016/j.antiviral.2014.04.006.
23. Xu D, Wang C, Zhu X, Zhao , et al. The antidepressant-like effects of fluvoxamine in mice involve the mTOR signaling in the hippocampus and prefrontal cortex. Psychiatry Res 2020;285: 112708 https://doi.org/10.1016/j.psychres.2019.112708.
24. Martin RJ, Robertson AP, Choudhary S. Ivermectin: An anthelmintic, an insecticide, and much more. Trends in Parasitol 2021;37(1):48-64 https://doi.org/10.1016/j.pt.2020.10.005.
25. Duru CE, Duru IA, Bilar A. Computational investigation of sugar fermentation inhibition by bergenin at the pyruvate decarboxylate isoenzyme 1 target of Scharomyces cervisiae. Journal of Med Plants Stud 2020;8(6):21-25 https://doi.org/10.22271/plants.2020.v8.i6a.1225.
26. Enenebeaku UE, Duru CE, Mgbemena IC, Ukwandu NCD, Nwigwe NH, Enenebeaku CK, Okotcha EN. Phytochemical evaluation and molecular docking of bioactive compounds from the roots of Dictyandraarborescens (Welw.) against Plasmodium bergheiprotein targets. Trop J Nat Prod Res 2021;5(2):370-381 https://doi.org/10.26538/tjnpr/v5i2.27.
27. Stojanovic SÐ, Zaric SD. Hydrogen bonds and hydrophobic interactions of porphyrins in porphyrin-containing proteins. Open Struct Biol J 2009;3(1):34-41 https://doi.org/10.2174/1874199100903010034.
Figure 1.Human ACE2 protein showing interacting amino acid residues (i.e. 19, 24, 28, 30, 31, 34, 38, 41, 42, 78, 82, 83) at ACE2-spike protein complex interface. 
Figure 2.Corona virus spike glycoprotein showing interacting amino acid residues (i.e 353, 355, 417, 446, 449, 453, 475, 484, 486, 478, 489, 496, 498, 500, 502) at ACE2-Spike protein complex interface. 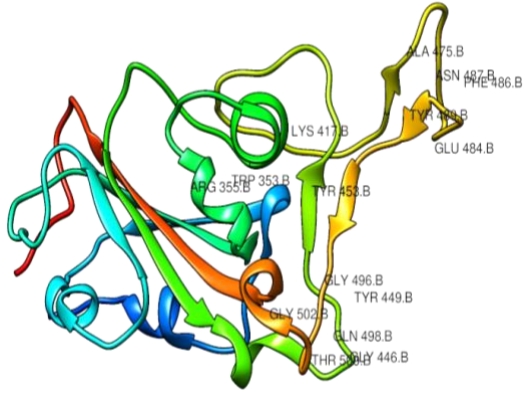
Figure 4.Interactions of A. Ivermectin B. Nafamostat C. Camostat with amino acids on spike glycoprotein. 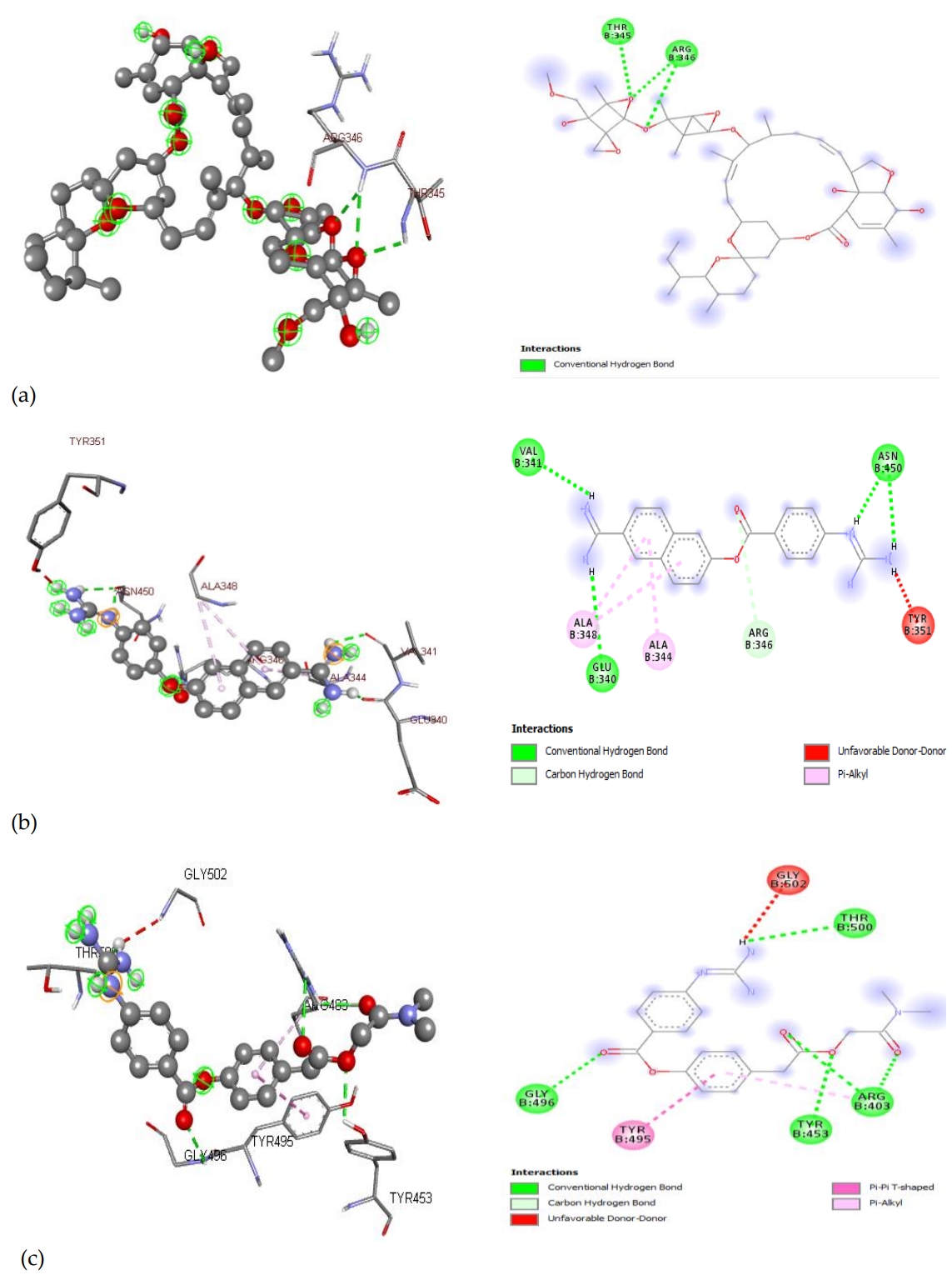
Table 1.Binding affinities of drug compounds on the human ACE2 and spike glycoprotein of COVID-19. Table 2.Amino acid residues involved in the interaction with drugs and human ACE2 protein. Table 3.Amino acid residues involved in the interaction with drugs and spike glycoprotein. |
|
|||||||||||||||||||||||||||||||||||||||